As solar and storage gain more momentum in the public eye, more questions about the current and future battery technologies are inevitable. Although there are many promising battery chemistries and technologies, as well as the tried-and-true lead-acid variants, this article focuses on lithium since it is the dominant player in global residential, commercial, and utility-scale storage.

Lithium-ion
Lithium-ion (Li-ion, or LI) batteries have been around since the '70s and are used in many consumer electronics, e-bikes and e-scooters, and electric cars and trucks. They have a higher energy density than traditional batteries, which means you can pack more power into a smaller footprint, like cramming 10 pounds of peanuts into a 5-pound bag. Li-ion batteries do not suffer (much) from “memory” losses which means they can sit fat, dumb and happy at any point in their charge cycle and recover to a 100% state of charge. By the way, this memory effect is only applicable to the nickel-cadmium batteries from the '70s and 80s but somehow applied to every other battery on the market, even to this day. It is a myth, even lead acid doesn't suffer from it.
There are many types of lithium-ion batteries, each engineered with their own different materials. When digging into the various kinds of Li-ion batteries, it seems that battery chemists are having fun playing around with different kinds of metals to see how each one reacts. The goal is to cram as many peanuts into the smallest bag possible; however, this process does have limitations. You never get something for nothing.
A quick Internet search reveals Li-ion battery types by their chemical designation: LiNixMnyCozO2, LiFePO4, and LiNiCoAlO2 to name a few. To the laymen, it is an incoherent jumble of the periodic table, but the engineers were quite clever in combining these materials together to take advantage of the strengths of each element.
There are five or six main types of Li-ion batteries. The most common ones used in residential storage applications - Nickel Manganese Cobalt Oxide (NMC) and Lithium Iron Phosphate (LFP) are explained below.
NMC
Nickel Manganese Cobalt Oxide (NMC) batteries use nickel as the primary metal on the battery cathode. Nickel, if used by itself, has a high energy rating but is unstable. So, engineers mix in a little manganese because it has low internal resistance. Presto! Now we have a battery that is more stable with high specific energy (capacity). Think Merlin, but with better access to exotic materials.
|
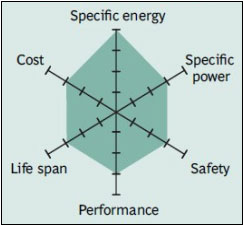
Most consumer electronics with rechargeable batteries use Cobalt Oxide because it has a high specific energy density. We need our cell phones to last as long as possible while we are texting people while out to dinner with our significant others. Unfortunately, this chemistry is very expensive, and using them for large storage applications may not be economically feasible- but that doesn’t stop people from trying. |
 |
NMC chemistry is used in a lot of consumer handheld electronics and in electric vehicles because it has a higher power density than other chemistries. The Nissan Leaf uses NMC and does the Tesla EV’s. However, using this chemistry in a stationary storage application will likely yield varying results in battery longevity and cycle count since the batteries are engineered for EV’s. You never get something for nothing.
Generally speaking, NMC is on the low end of the Li-ion family with regards to safety. This does not mean the chemistry is unsafe, it just means the next chemistry I will talk about is safer. It’s like comparing a car with just seat belts to one with seat belts, anti-lock brakes, and airbags. One care is safer than the other.
LFP
|
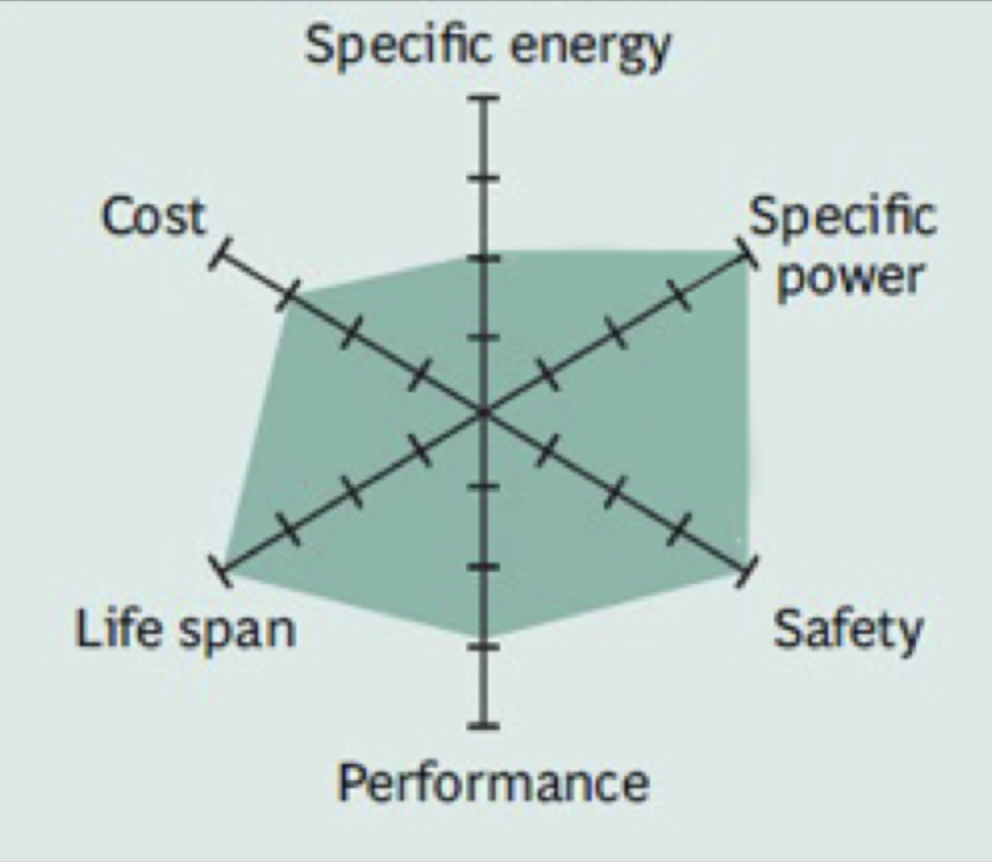
The Iron Phosphate battery (LiFePo2) uses phosphate as a stabilizer for one of the highest safety ratings of the family and for its good performance when used in a stationary battery application. Plus, LiFePo is easy to say and remember. This is the chemistry the Tigo EI Battery, and most other battery manufacturers use for exactly these reasons. Sure, Iron Phosphate doesn’t have the obscene energy density other Li-ion batteries have, but it is still pretty good on capacity and very safe. Ask me how I know… |
 |
Understanding lithium-ion battery charging may be challenging, especially if you are used to lead-acid batteries used with a charge controller. Before I left SMA, Li-ion batteries had only just begun to be supported by the Sunny Island. By “supported” I mean that there was a firmware addition that basically disengaged the Sunny Island charge controller and let the Li-ion manufacturer's Battery Management System (BMS) take over. This is a good thing since battery inverters and charge controllers typically charged the battery bank based on the voltage (among other things) at their positive and negative terminals. Li-ion batteries are charged very differently; each cell is monitored independently and the BMS must make micro changes to the charging of each cell.
Here is an animated example of a single EV battery bank cell showing the reversal of ions as the cell is charged and discharged. The anode and cathode (in the traditional sense) actually change positions depending on whether the cell is charging or discharging. Cool!

Speaking of cell voltages, Li-ion cells are kept at a higher voltage than their lead-acid cousins- typically about 3.3 to 3.7V per cell. The BMS behaves a little differently (in general) than the lead-acid chargers. First, the BMS applies a constant current while increasing the voltage. The BMS must then balance out the voltages across each individual cell. Some chargers can do this very quickly and skip this phase (superchargers). Once the cells are balanced and the voltages are in spec, the BMS just has to babysit the bank- one cell at a time- by applying a constant voltage.
When using Li-ion BMS in conventional battery backup systems and/or self-consumption applications, the system runs in virtual cruise control with little intervention required by the homeowner. In a time of use application, the peak and off-peak windows are set each year as the windows change. Additionally, Li-ion batteries are maintenance-free and do not require a lot of attention. The less time the homeowner spends watching the system the better. The BMS will let you know if something has gone wrong. Setting cell float voltages, absorption time, and other lead-acid parameters are a thing of the past.
That is the end of this 30,000-foot overview. Do you have any questions? Let’s hear them! Do you want to geek out some more on batteries and future technologies? Let’s do it!